CORMEDIX INC. REPORTS FIRST QUARTER FINANCIAL RESULTS AND PROVIDES BUSINESS UPDATE
- Anticipate Data Safety Monitoring Board review of interim analysis in July 2018; review was triggered in December 2017 with 28 confirmed cases of catheter-related bloodstream infections (CRBSIs) identified in the Phase 3 LOCK-IT-100 trial
- Granted U.S. FDA orphan drug designation for taurolidine for treatment of neuroblastoma
- Conference Call Today at 4:30 p.m. Eastern Time
Listen to Conference Call:
Berkeley Heights, NJ – May 15, 2018 – CorMedix Inc. (NYSE American: CRMD), a biopharmaceutical company focused on developing and commercializing therapeutic products for the prevention and treatment of infectious and inflammatory disease, today announced financial results for the first quarter ended March 31, 2018 and provided an update on recent business highlights.
“We are continuing to make progress on our data collection efforts for our Phase 3 LOCK-IT 100 study, which is ongoing,” said Khoso Baluch, Chief Executive Officer of CorMedix. “Concurrent to the work on the interim analysis, we continued to progress the Phase 3 trial, having enrolled approximately 790 subjects, exceeding our original enrollment target of 632 patients. The additional trial subjects should help to bring us closer to our goal of achieving the requisite number of catheter-related blood stream infection (“CRBSI”) events in the Phase 3 study. We are working diligently to capture, investigate and prepare clinical packages for review by the Clinical Adjudication Committee (“CAC”) as quickly as possible.”
Mr. Baluch continued, “We remain completely dedicated to the successful completion of the Neutrolin® registrational program, and to bringing Neutrolin to market in the U.S. We believe Neutrolin has the potential to meaningfully improve patient care by significantly reducing or eliminating costly and potentially deadly catheter-related bloodstream infections.”
“With respect to our taurolidine asset, we were pleased to receive orphan drug designation during the first quarter for taurolidine in the treatment of neuroblastoma. We received the results of the preliminary animal studies in taurolidine-infused sutures, meshes, and hydrogels and currently anticipate that any future development will be dependent on receipt of marketing approval of Neutrolin in the U.S.,” Mr. Baluch added. “We continue to evaluate our capital needs and options and are committed to ensuring the company has sufficient cash on hand to complete the interim analysis, with a near-term financial goal of minimizing dilution to our shareholders.”
Recent Corporate and Clinical Highlights:
- Reviewed and source-verified 54% of the LOCK-IT 100 data. None of the 28 CAC-reviewed CRBSI cases are affected by this additional review. On track to complete the review and source-verification efforts by the end of the second quarter 2018.
- Completed the animal studies of taurolidine-infused sutures, meshes and hydrogels. Further development primarily dependent on U.S. marketing approval of Neutrolin.
- Added Dr. Paul Chew as Chief Medical Officer consultant and advisor to support completion of the Company’s Neutrolin clinical program and to further the neuroblastoma and medical device pipeline.
- Enrolled approximately 790 subjects in its multi-center Phase 3 registrational trial for Neutrolin in hemodialysis patients with central venous catheters.
- Granted orphan drug designation by the Food and Drug Administration for taurolidine in the treatment of neuroblastoma.
First Quarter 2018 Financial Highlights
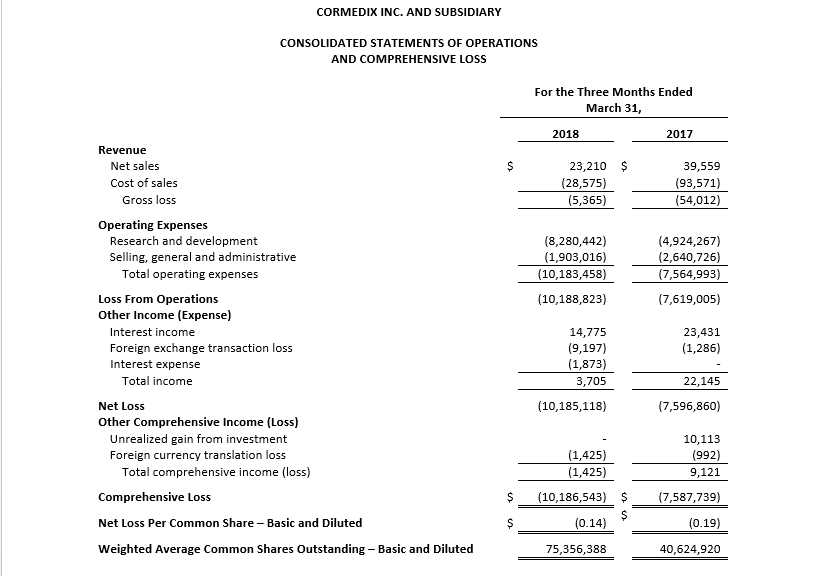
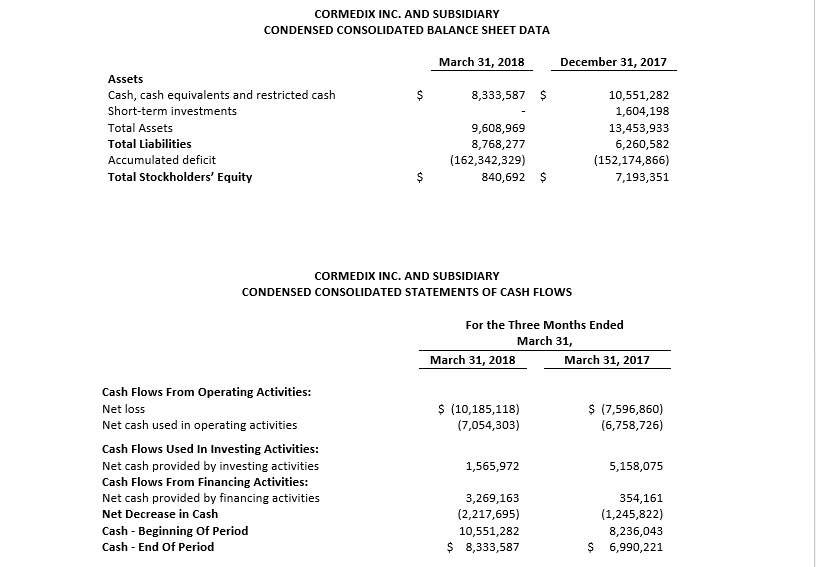
For the first quarter 2018, CorMedix recorded a net loss of $10.2 million, or $0.14 per share, compared with a net loss of $7.6 million, or $0.19 per share, in the first quarter of 2017, an increase of $2.6 million. Net loss in the first quarter of 2018 was driven by increased costs related to the ongoing LOCK-IT-100 clinical study while other expenses continued to decline or remained flat.
Operating expenses in the first quarter 2018 were $10.2 million, compared to $10.4 million in the fourth quarter of 2017, a decrease of approximately 2%. R&D expense decreased approximately 2% to $8.3 million, due to a 77% decrease in manufacturing expense and a 29% decline in other R&D, while clinical trial expense increased by 21%. SG&A expense declined by 3%. Approximately 71% of total operating expenses during the first quarter of 2018 were related to the LOCK-IT 100 clinical trial.
Conference Call Information:
CorMedix CEO, Khoso Baluch will host a conference call and webcast today, May 15, 2018, at 4:30 PM Eastern Time, to discuss recent corporate developments and financial results. Call details and dial-in information is as follows:
Tuesday, May 15 at 4:30pm Eastern Time
Domestic: 877-407-9124
International: 201-689-8584
Conference ID: 13679901
Webcast: http://www.investorcalendar.com/event/29615
A replay will be available through May 28 by dialing:
Domestic: 877-481-4010
Replay PIN: 29615
About CorMedix
CorMedix Inc. is a biopharmaceutical company focused on developing and commercializing therapeutic products for the prevention and treatment of infectious and inflammatory diseases. The Company is focused on developing its lead product Neutrolin®, a novel, non-antibiotic antimicrobial solution designed to prevent costly and dangerous bloodstream infections associated with the use of central venous catheters, currently in a Phase 3 clinical trial enrolling patients undergoing chronic hemodialysis. Such infections cost the U.S. healthcare system approximately $6 billion annually and contribute significantly to increased morbidity and mortality. Neutrolin has FDA Fast Track status and is designated as a Qualified Infectious Disease Product, which provides the potential for priority review of a marketing application by FDA and allows for 5 additional years of QIDP market exclusivity in the event of U.S. approval. Neutrolin is already marketed as a CE Marked product in Europe and other territories. In parallel, CorMedix is leveraging its taurolidine technology to develop a pipeline of antimicrobial medical devices, with active programs in surgical sutures and meshes, and topical hydrogels. The company is also working with top-tier researchers to develop taurolidine-based therapies for rare pediatric cancers. For more information, visit: www.cormedix.com.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, including with respect to possible uses of taurolidine, that are subject to risks and uncertainties. All statements, other than statements of historical facts, regarding management’s expectations, beliefs, goals, plans or CorMedix’s prospects, future financial position, financing plans, future revenues and projected costs should be considered forward-looking. Readers are cautioned that actual results may differ materially from projections or estimates due to a variety of important factors, including: the cost, timing and results of the ongoing and planned Phase 3 trials for Neutrolin® in the U.S. and the resources needed to commence and complete those trials, including the interim analysis for the ongoing Phase 3 clinical trial; the risks and uncertainties associated with CorMedix’s ability to manage its limited cash resources and the impact on current, planned or future research, including the ongoing LOCK-IT 100 trial and research for additional uses for taurolidine; obtaining additional financing to support CorMedix’s research and development and clinical activities and operations; the possible inability to capture sufficient CRBSI events in the ongoing Phase 3 clinical trial for Neutrolin®; preclinical results are not indicative of success in clinical trials and might not be replicated in any subsequent studies or trials; and the ability to retain and hire necessary personnel to staff our operations appropriately. These and other risks are described in greater detail in CorMedix’s filings with the SEC, copies of which are available free of charge at the SEC’s website at www.sec.gov or upon request from CorMedix. CorMedix may not actually achieve the goals or plans described in its forward-looking statements, and investors should not place undue reliance on these statements. CorMedix assumes no obligation and does not intend to update these forward-looking statements, except as required by law.
Investor Contact:
Dan Ferry
Managing Director
LifeSci Advisors
617-535-7746